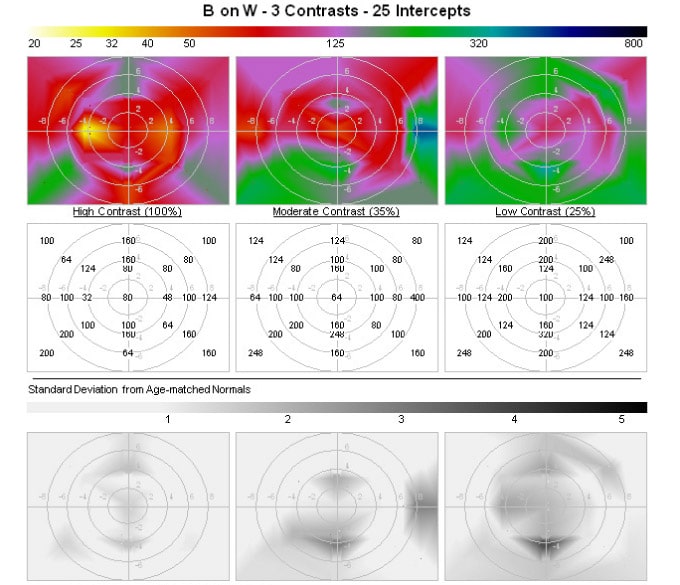
The Omnifield, is the first device to measure functional resolution throughout the central 20° diameter visual field (functional field of view) under conditions of luminance and contrast that mimic those required for vision tasks that are encountered during common day and evening activities. When testing is performed with the Omnifield the patient is seated 1 meter in front of a large monitor (figure 1) and after an introductory episode wherein they are advised to fixate on the center of a large red cross, testing is performed in which Landolt C’s are flashed for 0.1 to 0.3 seconds (centered within the cross center on a pedestal and at standard loci within the central 10 degree radius field. The C’s are randomly positioned with the opening facing up, down, right or left; the patient responds by deflecting a joystick. Sufficient time is allowed for the person to respond between presentations (recording and tracking the response times with program adjustment). The C’s are enlarged or shrunken until a threshold is reached, at each field locus. Fixation tracking is monitored at all times so that the C presentation field positions are always assured. Visual field testing for one or two photopic and/or mesopic environments including reproducibility requires 5-7 minutes, including reproducibility. The device is FDA approved for clinical supervised use and has been utilized in clinical trials and extensively validated, demonstrating earlier pathologic associations than obtained with chart acuity, Humphrey perimetry, or contrast sensitivity, when testing is conducted under mesopic environments with reduced contrasts, paralleling patient reported complaints (via a self-administered, electronic visual function questionnaire, also developed by Sinclair Technologies LLC), and with ERG abnormalities.