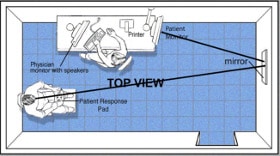
The Central Vision Analyzer (CVA), developed by Sinclair Technologies,
is the first clinical device to measure monocular or binocular resolution
capabilities at the center of vision under conditions of luminance and
contrasts that mimic those encountered in the real world (please also review the Omnifield device). In addition to standard electronic charts utilized for refraction the CVA also offers an interactive testing in which round C’s are flashed on the viewed screen in one of 4 positions (opening up, down right, or left) with presentation times that are standard of fixations measured in common tasks (0.1-0.3 secs) and with sufficient time allowed for the person to respond by pressing a joystick in the same direction as the opening (show figure). A true psychophysical threshold is achieved utilizing steps in between those of the ETDRS chart, and testing is pursued in modules selected for up to 6 luminance and contrast conditions that mimic real world tasks (i.e. restaurant dining, evening driving, playing golf or tennis in the sun) requiring approximately 2-3 minutes for testing both eyes including reproducibility.
The CVA has been used to evaluate real-world functional vision in eyes with many types of pathology and under therapeutic trial outcomes; cataracts pre and post surgery with various IOL’s implanted, patients with AMD and HIV disease with and without retinopathy or in eyes of diabetics with retinopathy and macular edema undergoing a number of treatments. It has been used in screening settings including Special Olympics, in which it performed well in children of all ages with mental difficulties, correlating better with the diagnosed ocular pathologies than chart measurements. The CVA is FDA approved for use under a practitioner’s supervision. Current work is being pursued to develop this test tint evaluation for sports participation as well as presentations on smart phones to enable patient monitoring at home with much improved sensitivity in monitoring disease progression.
The Central Vision Analyzer (CVA), developed by Sinclair Technologies,
is the first clinical device to measure monocular or binocular resolution
capabilities at the center of vision under conditions of luminance and
contrasts that mimic those encountered in the real world (please also review the Omnifield device). In addition to standard electronic charts utilized for refraction the CVA also offers an interactive testing in which round C’s are flashed on the viewed screen in one of 4 positions (opening up, down right, or left) with presentation times that are standard of fixations measured in common tasks (0.1-0.3 secs) and with sufficient time allowed for the person to respond by pressing a joystick in the same direction as the opening (show figure). A true psychophysical threshold is achieved utilizing steps in between those of the ETDRS chart, and testing is pursued in modules selected for up to 6 luminance and contrast conditions that mimic real world tasks (i.e. restaurant dining, evening driving, playing golf or tennis in the sun) requiring approximately 2-3 minutes for testing both eyes including reproducibility.
The CVA has been used to evaluate real-world functional vision in eyes with many types of pathology and under therapeutic trial outcomes; cataracts pre and post surgery with various IOL’s implanted, patients with AMD and HIV disease with and without retinopathy or in eyes of diabetics with retinopathy and macular edema undergoing a number of treatments. It has been used in screening settings including Special Olympics, in which it performed well in children of all ages with mental difficulties, correlating better with the diagnosed ocular pathologies than chart measurements. The CVA is FDA approved for use under a practitioner’s supervision. Current work is being pursued to develop this test tint evaluation for sports participation as well as presentations on smart phones to enable patient monitoring at home with much improved sensitivity in monitoring disease progression.